 So we could say that Charles' Law describes how hot air balloons get light enough to lift off, and why a temperature inversion prevents convection currents in the atmosphere, and how a sample of gas can work as an absolute thermometer. So the net effect is that the pressure doubles if the container doesn't stretch, or the volume doubles if the container enlarges to keep the pressure from rising. or VkT, where k is the proportionality constant. Consider a gas at pressure P and temperature T having volume V, then Charless law states that : VT. Specifically, if we double the Kelvin temperature of a rigidly contained gas sample, the number of collisions per unit area per second increases by the square root of 2, and on average the momentum of those collisions increases by the square root of 2. Charless law states that at constant pressure, the volume of a given mass of an ideal gas is directly proportional to its temperature. Inside a helium balloon, about 10 24 (a million million million million) helium atoms smack into each square centimeter of rubber every second, at speeds of about a mile per second!īoth the speed and frequency with which the gas molecules ricochet off container walls depend on the temperature, which is why hotter gases either push harder against the walls (higher pressure) or occupy larger volumes (a few fast molecules can occupy the space of many slow molecules). They push outward on flasks or pistons or balloons simply by bouncing off those surfaces at high speed. Under typical conditions, gas molecules are very far from their neighbors, and they are so small that their own bulk is negligible. The accepted explanation, which James Clerk Maxwell put forward around 1860, is that the amount of space a gas occupies depends purely on the motion of the gas molecules. It is pretty surprising that dozens of different substances should behave exactly alike, as these scientists found that various gases did. In fact, Guillaume Amontons had done the same sorts of experiments 100 years earlier, and it was Joseph Gay-Lussac in 1808 who made definitive measurements and published results showing that every gas he tested obeyed this generalization. The irony is that Charles never published the work for which he is remembered, nor was he the first or last to make this discovery. The law's name honors the pioneer balloonist Jacques Charles, who in 1787 did experiments on how the volume of gases depended on temperature. The physical principle known as Charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the Kelvin scale (zero Kelvin corresponds to -273.15 degrees Celsius). Lindeman, professor and chair of the chemistry department of Colorado College in Colorado Springs, offers this explanation: Closer to the condensation point, the linear relationship does not hold up volume decreases more rapidly than temperature.Theodore G. 
Real gas *es behave in accordance with Charles' Law at temperatures well above the gas' condensation point. While Charles' Law describes the behavior of ideal gases, not real ones, the law does have real-world applications. The relationship is linear, if the temperature of a volume of gas doubles, the volume doubles. For this relationship to hold, both the mass of the gas and its pressure are held constant, and the temperature must be reported in Kelvin. Where V 1 is the volume of the gas at one temperature (T 1) and, V 2 is the volume after a change to a new temperature (T 2). 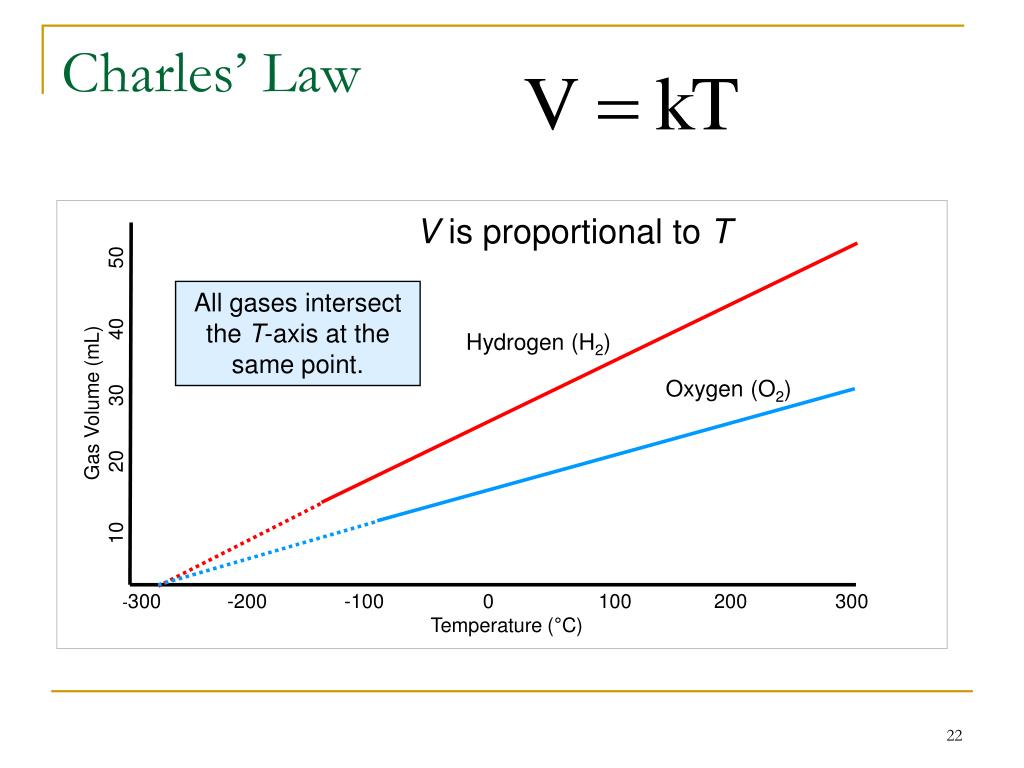
This relationship allows changes in the volume of a fixed mass * of gas to be calculated given a change in temperature. However, if the container volume is adjustable, the volume will increase, and the pressure will remain the same.Ĭharles' Law is the formal description of this relationship between temperature and volume at a fixed pressure. In a rigid container, the more frequent and forceful collisions result in higher pressure. As the molecules move faster, they encounter the walls of the container more often and with more force. Increasing the temperature of a volume of gas causes individual gas molecules to move faster. 
In a rigid, but adjustable container such as a sealed syringe, the collisions of the moving gas molecules with the syringe walls provide the force that resists efforts to move the syringe plunger, creating pressure inside of the syringe. In a balloon, the force of individual molecules hitting the inside of the balloon keeps the balloon inflated. When this happens, Newton's Third Law of motion says that both the molecule and the wall will experience a force. When a molecule encounters a wall, it bounces off and moves off in a different direction. 
The molecules that make up a gas move in straight lines until they encounter another molecule or the walls of a container. This illustration explores the relationship between the temperature and volume * of an ideal gas * in a container that adjusts to allow pressure to remain constant. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
